Improved Assays with Magnetically Coated
3D Cell Models
Introduction
In recent years, the need to have physiologically accurate 3D cell models for research and drug development has been steadily growing. Researchers have been perfecting the formation and maintenance of various 3D models for understanding both disease and normal physiology1,2. Some of the limiting factors have been the ability to perform complex assays easily and quickly with these precious samples especially patient-derived material. Also, when performing drug treatments and assays in multi-well plates one is limited by the number of readouts per sample. The manual treatment, staining, and processing of spheroids and organoids is typically labor-intensive and prone to disruption or loss of samples. In addition, high content imaging can be problematic because organoids tend to locate at the edges of wells.
To overcome these issues during assays, we demonstrate the use of our Pu·MA System® 3D MAG and 3D Flowchips to perform automated assay steps with magnetically coated 3D cell models (Figure 1). NanoShuttle™ coated 3D cell models were transferred and centered into flowchip wells using magnets. The cell models were unaffected during the assays and easily imaged using a confocal microscope.
- Media Sampling for secreted factors: Mouse islets were incubated with different glucose levels. Supernatants were collected in situ and analyzed for insulin secretion using ELISA.
- Drug treatment and staining: HeLa spheroids were treated with compounds for 24−48 hrs. Automated viability staining was used to determine concentration response curves.
- Immunofluorescence (IF) staining for biomarkers: TNBC patient-derived organoids4 were assayed for drug response. PDOs were stained with IF markers and imaged in the flowchips.
Pu·MA System 3D MAG Workflow
Pu·MA System and 3D flowchips have been designed for streamlined automated organoid assays. Pu·MA System 3D MAG consists of the Pu·MA System with the 3D MAG modification which enables magnetically coated 3D cell models to be held within the protected sample chamber during the assay and subsequent imaging (Figure 1 & 2). The NanoShuttle treatment of formed organoids was done as per protocols from Greiner Bio-One3. In this application note we demonstrate a novel use of this bioprinting process coupled to our automated assay system. The magnetized 3D cell models are transferred into the flowchips with the help of a loading tray which ensures centering of the organoids (Figure 2). This has been designed specifically for use with our flowchips. The flowchips are then placed into the system within the incubator. The system architecture of the Pu·MA System and use of pneumatics to move fluids provides gas exchange to the sample chambers within the incubator environment.
The flowchips are in a convenient multi-well plate format (384-well spacings SLAS/ANSI standard) which makes them amenable to multichannel or automated liquid dispense system. They have optically clear bottoms for imaging with any fluorescence or confocal imaging system (Figure 1).
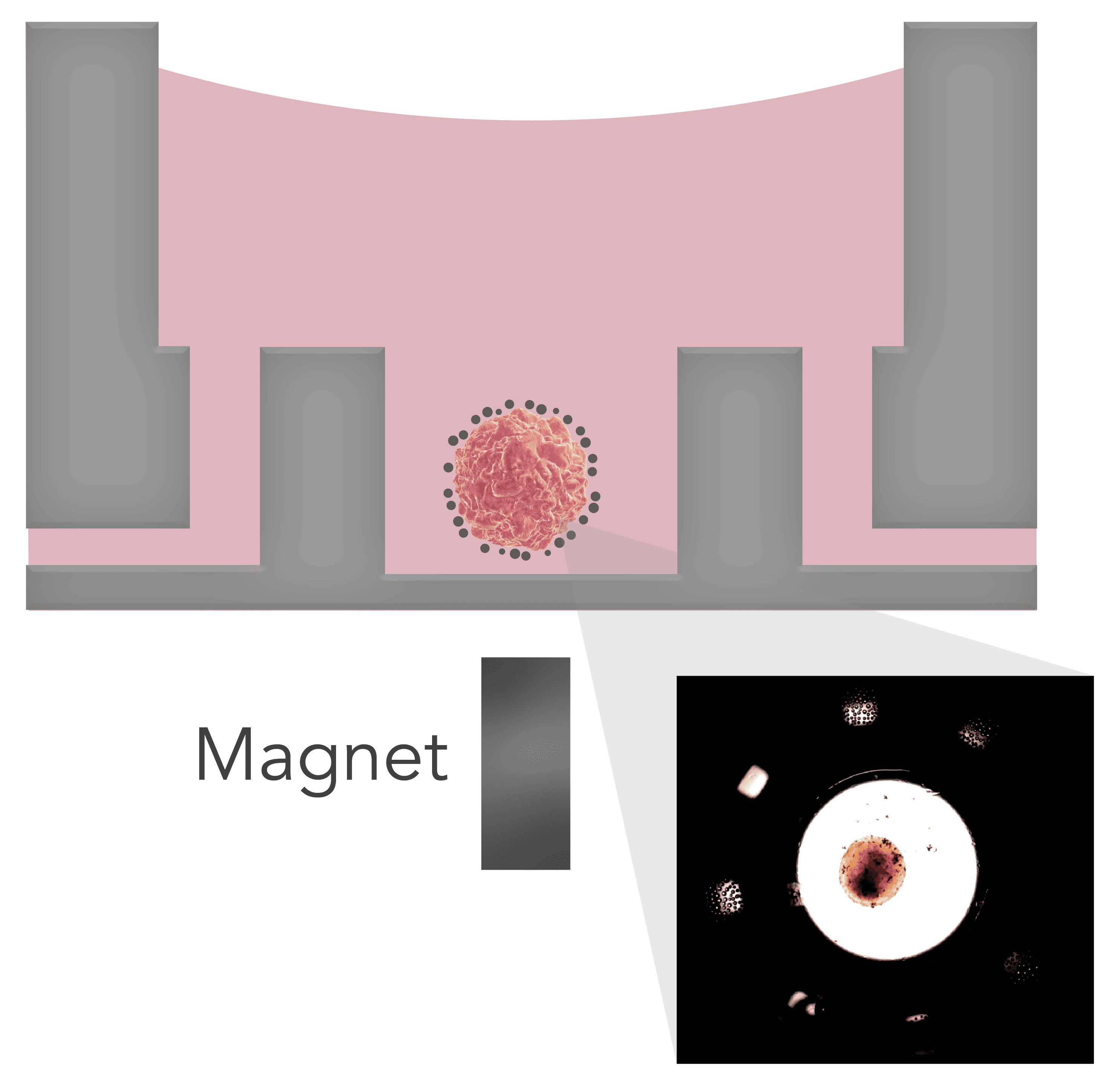
Figure 1. Schematic of a positioned spheroid within flowchip protected sample chamber in a Pu·MA System 3D MAG. Inset shows a bright field image of Nanoshuttle coated mouse islets within the sample chamber
