Transfer, Automated Staining, and Imaging of Organoids in Matrigel
Introduction
Organoid technology attracts an enormous interest for personalized medicine, drug screening and cell therapy 1 . In most cases, organoid formation is dependent on extracellular matrices (ECM). Matrigel is an ECM-based hydrogel matrix, that is often used to generate organoids. It provides physical support and molecules that are important for cell function, attachment, and differentiation 2 . Assays with fragile organoid structures rely on manual handling which limits the ability to perform complex assays easily, quickly, and reliably. Automation of the assay workflow using Pu·MA System® eliminates the need for labor intensive manual processing of organoids which is normally prone to disruption or loss of sample 3 . It enables the streamlining of complex multi-step procedures like drug treatment and immunofluorescence staining for imaging.
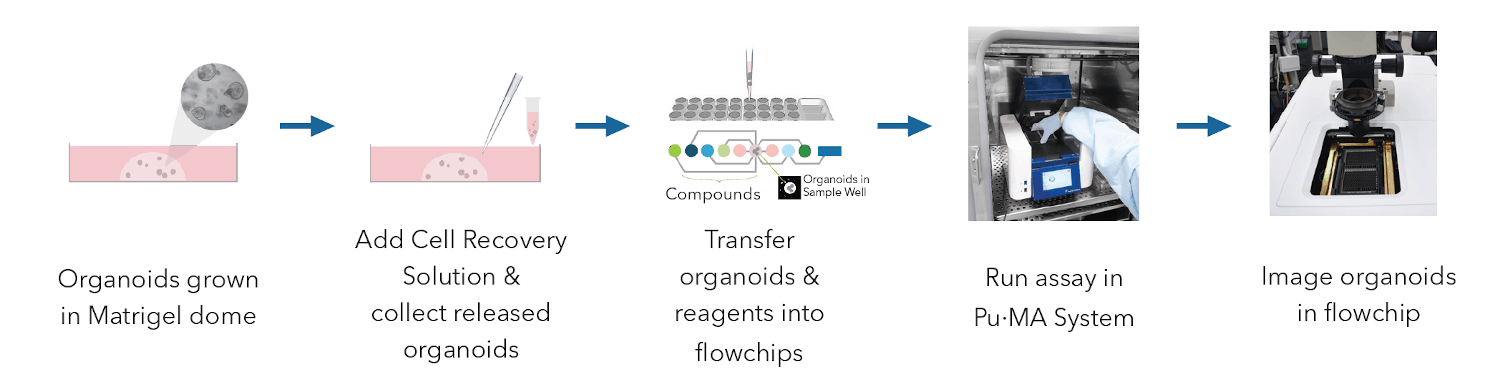
This technical note describes the procedure of retrieving pulmonary organoids from a Matrigel dome and transferring them to a Flowchip in 25% Matrigel to perform automated assays using Pu·MA System. A concentration range of 20% to 30% Matrigel was experimentally determined as optimal to support and maintain organoids inside the protected chamber of the flowchip. This concentration had no effect on the performance of microfluidic channels and liquid transfers within the flowchip. Transferred organoids were stained for viability using a Pu·MA System and imaged within the flowchip.
Procedure
In this protocol we used pulmonary organoids that were derived from primary human lung epithelial cells (ScienCells, Co.). They were grown in Matrigel domes using reagents and protocol from StemCell Technologies. Cells were expanded in 2D, mixed with GF reduced 90% Matrigel and seeded into Matrigel domes in 24-well plates. The workflow of this technote is shown in Figure 1 and the detail of steps are described below.